
Hardness: 4 – 4.5
D: 2.13 gm/cm3.
Luster: vitreous, pearly on {010}.
Streak: white
Biaxial (-).
α = 1.485, β = 1.496, γ = 1.498, δ = 0.013, 2Vx = 45°. a = X, b = Y, c = Z.
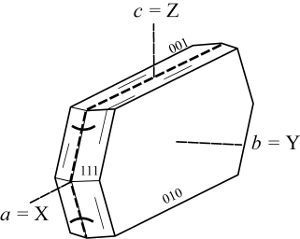
a 13.600, b 18.222, c 17.863 Å.
Z = 2, Space group Fmmm
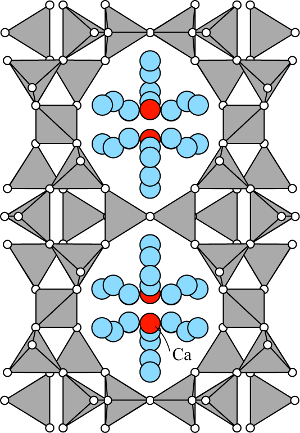
The framework topology of stellerite is the same as stilbite (STI), and with mostly Ca in the channels is orthorhombic, space group Fmmm (Galli and Alberti 1975, Miller and Taylor 1985). The neutron diffraction data of Miller and Taylor (1985) disclosed slight (Si,Al) ordering with individual tetrahedral Al contents ranging from 10 to 30%.
Stellerite has only one fully, or nearly fully occupied, Ca channel cation site (red) (Galli and Alberti 1975, Miller and Taylor 1985). Ca is coordinated only to channel H2O molecules (blue) and not to framework oxygens. Similar to stilbite, the Ca(H2O) complex is hydrogen bonded to the framework oxygens (Miller and Taylor 1985). There are seven partially occupied H2O sites in the structure with occupancies between 0.2 and 0.8 (Galli and Alberti 1975, Colella and Gualtieri, 2007).
Several of the known stellerite occurrences are in cavities of basalt. The type locality on Copper Island, Commander Islands (Kommandorskiye Ostrova), Russia, Bering Sea, is in brecciated basaltic rocks. Galli and Passaglia (1973) describe stellerite from lower Miocene andesitic breccia, exposed near Villanova Monteleone, Sardinia, Italy. Large crystals of stellerite occur in the Garrawilla Volcanics (Jurassic flows interbedded in sandstone), New South Wales, Australia (England and Sutherland 1988). Exceptional specimens have been found at Jalgaon, Maharashtra, India. There are no clear indications of why stellerite crystallized in these localities rather than stilbite-Ca, except for the lack of Na.
Hydrothermal systems.
The occurrences of stellerite in plutonic and metamorphic rocks are so similar to those of stilbite-Ca that paragenesis cannot be used to distinguish the two minerals. Like stilbite, stellerite occurs in some hydrothermal ore veins. One notable example is east of Kuruna, Sweden, at the old copper mine next to the Leveäniemi mine. This is the locality of the mineral, for which the name zeolite was first coined (Cronstedt 1756, Colella and Gualtieri 2007).
Cronstedt, A.K. 1756. [Observation and description of an unknown kind of rock to be named zeolites] Kongl. Vetenskaps Acad. Handl. Stockholm. 17, 120-3 (in Swedish).
Colella, C. and Gualtieri, A.F. 2007. Cronstedt’s zeolite. Microporous Mesoporous Mater. 105, 213-221.
England, B.M., Sutherland, F.L. 1988. Volcanic zeolites and associated minerals from New South Wales Min. Rec., 19, 389-406.
Galli, E. and Alberti, A. 1975. The crystal structure of stellerite. Bull. Soc. fr. Minéral. Cristallogr. 98, 11-18.
Miller, S.A. and Taylor, J.C. 1985. Neutron single-crystal diffraction study of an Australian stellerite. Zeolites, 5, 7-10.
Morozewicz, J. 1909. Über Stellerit, ein neues Zeolithmineral. Bull. International de l’Académie des Sciences de Cracovie, 344-359.
Neumann, H. 1944. Silver deposits at Kongsberg. Norg. Geol. Unders. 162, 1-133
Passaglia, E., Galli, E., Leoni, L. and Rossi, G. 1978. The crystal chemistry of stilbites and stellerites. Bull. Minéral. 101, 368-375.
Pelloux, A. 1949. Sopra alcune zeoliti della Valle del Varenna presso Pegli con descrizone di un minerale nuovo per l’Italia. Ann. Mus. Civ. Stor. Nat. Genova, 63, 1-8.
Saebø, P.C., Reitan, P.H., and Gaul, J.J.C. 1959. Stilbite, stellerite and laumontite at Honningvag, Northern Norway. Norg. Geol. Unders., 205, 171-173.
Young, M. 1965. Stellerite and its genesis in a disseminated molybdenum ore. Sci. Geol. Sin. 3, 295-302.