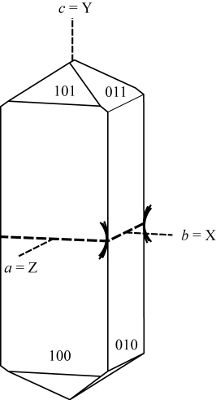
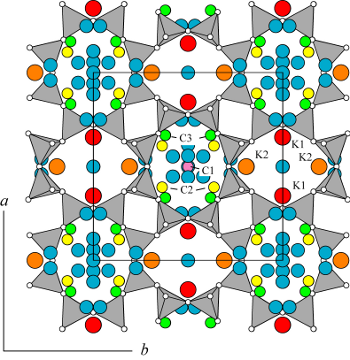
| |
Merlinoite has been reported from very few localities, in part because it is so easily mistaken for phillipsite. Its occurrence in basaltic deep sea sediment, in the saline and alkaline Searles Lake, California, and in nepheline basalt cavities shows that special chemical environments are necessary for crystallization.
Diagenesis of sediment and sedimentary rocks. Merlinoite has been identified in deep sea sediment or nodules in two different settings. It was first found in manganese nodules in the Indian Ocean (Mohapatra and Sahoo 1987), where it replaces pyroclastic fragments. Merlinoite occurs with smectite and phillipsite in pore spaces of Late Miocene vitric sandstone at a depth of 171 m below the sea floor at Site 841 (Ocean Drilling Program) at the Tonga arc edge (Vitali et al. 1995, Tazaki and Fyfe 1992).
Guided by experimental work that indicated merlinoite crystallizes readily from potassic, alkaline solutions, Donahoe et al. (1984) studied the zeolite in ashy layers in the sediment of Searles Lake, California, a playa lake deposit with potassic brines (Smith et al. 1983). They found merlinoite with phillipsite in two ash beds at the 41 and 42 m levels of the 915 m KM-3 drill core. Ash beds from deeper levels contain clinoptilolite, analcime, and K-feldspar (Hay and Guldman 1987). Energy dispersive spectra of coexisting merlinoite and phillipsite indicate that the Si/Al contents are about the same and that merlinoite has a higher K but lower Na content than phillipsite.
Cavities in basaltic lava. Merlinoite occurs in cavities of a few weakly altered, glassy alkaline-basaltic lavas. At the type locality, it occurs in veinlets cutting the kalsilite melilitite exposed in Cupaello Quarry near Santa Rufina, Rieti, Italy (Passaglia et al. 1977). Tiny spherical aggregates of merlinoite are associated with apophyllite, phillipsite, calcite, and chabazite. Della Ventura et al. (1993) describe the merlinoite occurring in ejecta from Fosso Attici near Sacrofano north of Rome. The merlionite here occurs as pseudotetragonal prisms up to 5 mm in length.
In their paper describing amicite as a new mineral Alberti et al. (1979) mention the associated merlinoite. These zeolites occur in veins cutting the Tertiary melilite-nepheline basalt and pyroclastic rock of the volcano at Höwenegg, Hegau, Germany. Merlinoite occurs in the tiny vesicles of a sintered sandstone xenolith in the alkali olivine basalt at the Ortenberg quarry, Vogelsberg, Hessen, (Hentschel 1986). It has also been found at the contact between basalt and sediments at Halbinsel Tjornes on the north coast of Iceland (Schnorrer-Köhler 1990).
Deuteric alteration. Khomyakov et al. (1981) report the occurrence of merlinoite in hydrothermally altered cataclastic pegmatoid rocks in the Vuonnemiok River valley, Khibina massif, Kola Peninsula, Russia. It occurs as irregular grains and subhedral equant crystals in association with pectolite, catapleiite, astrophyllite, lamprophyllite, fibrous lorenzenite, aegerine, and fluorite. |
| |
Alberti, A., Hentschel, G. Vezzalini, G. 1979. Amicite, a new natural zeolite. Neues Jahrb. Mineral., Mh., 1979, 481-488.
Deer, A., Howie, R., Wise, W.S., and Zussman, J. (2004). Rock Forming Minerals. vol. 4B.Framework Silicates: Silica Minerals, Feldspathoids and the Zeolites. The Geological Society, London.
Della Ventura, G., Parodi, G.C. and Burragato, F. 1993. New data on merlinoite and related zeolites. Rend. Linceri Sci. Fisiche Naturalli, Ser. 9, 4, 303-312.
Donahoe, R.J., Liou, J.G., and Guldman, S. 1984. Synthesis and characterization of zeolites in the system Na2O-K2O-Al2O3-SiO2-H2O. Clays and Clay Miner. 32, 433-443.
Galli, E., Gottardi, G., and Pongiluppi, D. 1979. The crystal structure of the zeolite merlinoite. Neues Jahrb. Mineral., Mh., 1979, 1-9.
Hay, R.L. and Guldman, S.G. 1987. Diagenetic alteration of silicic ash in Searles Lake, California. Clays and Clay Miner. 35, 449-457.
Hentschel, G. 1986. Paulingit und andere seltene Zeolithe in einem gefritteten Sandsteineinschluss im Basalt von Ortenberg (Vogelsberg, Hessen). Geol. Jahrb. Hessen 114, 249-256.
Khomyakov, A.P., Kurova, T.A. and Muravishkaya, G.I. 1981. Merlinoite, first occurrence in the USSR. Dokl. Acad. Sci. U.S.S.R., Earth Sci. Sect. 256, 172-174.
Mohapatra, B.K. and Sahoo, R.K. 1987. Merlinoite in manganese nodules from the Indian Ocean. Min. Mag. 51, 749-750.
Passaglia, E., Pongiluppi, D., Rinaldi, R. 1977. Merlinoite, a new mineral of the zeolite group. Neues Jahrb. Mineral., Mh., 1977, 355-364.
Schnorrer-Köhler, G. 1990. Merlinoite -- ein neues Zeolithmineral von der Halbinsel Tjornes, N. Island. Aufschluss 41, 295-298.
Smith, G.I., Barczak, V.J., Moulton, G.F., and Liddicoat, J.C. 1983. Core KM-3, a surface-to-bedrock record of late Cenozoic sedimentation in Searles Valley, California. U.S. Geol. Surv. Prof. Pap. 1256, 24 pp.
Tazaki, K. and Fyfe, W.S. 1992. Diagenetic and hydrothermal mineral alteration observed in Izu-Bonin deep-sea sediments, Leg 126. Proceedings of the Ocean Drilling Program, Scientific Results 126, 101-112.
Vitali, F., Blanc, G., and Larqué,P. 1995. Zeolite distribution in volcaniclastic deep-sea sediments from the Tonga Trench margin (SW Pacific). Clays and Clay Minerals 43, 92-104. |